First Cryo-EM structure of the human TRPV4 ion-channel in the open conformation, in complex with an agonist.
In partnership with Bayer, leadXpro has generated protein and solved the cryo-EM structure of human TRPV4 in complex with the agonist 4α-PDD, providing novel information about agonist binding to this important drug target.
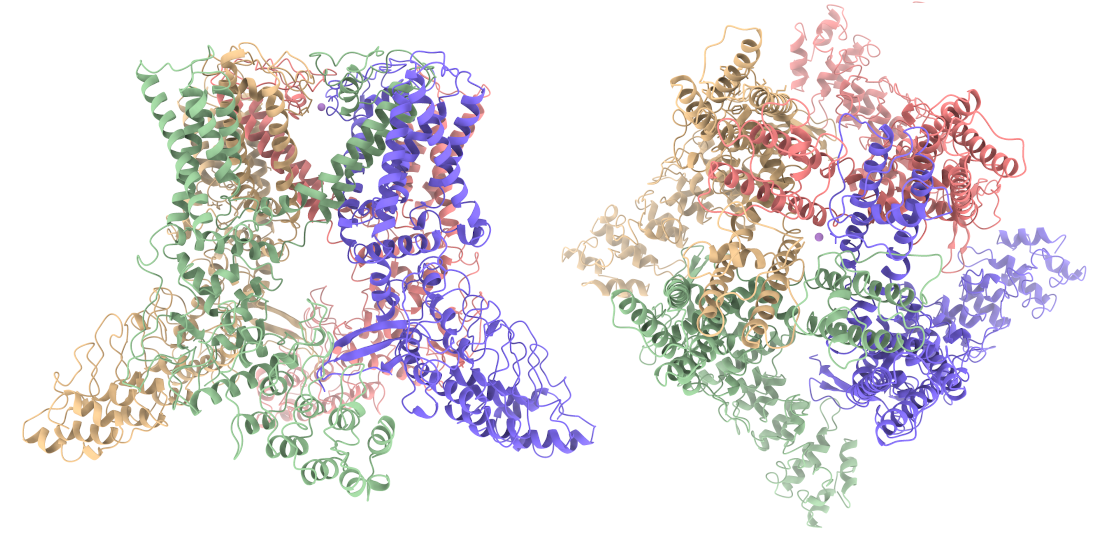
TRPV4 is a nonselective calcium channel that is involved in multiple physiological functions. The structure of TRPV4 in complex with 4α-PDD represents the first structural information of hTRPV4 in a ligand-induced open conformation. Complementary mutagenesis and functional assay experiments support the EM-identified binding site for 4α-PDD, and also allow the rationalisation of disruptive mutations located outside of the 4α-PDD binding site. Information about the agonist binding mode can now be used to discover and optimize novel drug candidates.
Reference:
Cryo-EM structural studies of the agonist complexed human TRPV4 ion-channel reveals novel structural rearrangements resulting in an open-conformation (2020-10-13).
DOI: 10.1101/2020.10.13.334797; https://www.biorxiv.org/content/10.1101/2020.10.13.334797v1.full.pdf