Enabling design of selective antagonists for the CC Chemokine receptor 2A (CCR2A)
In a partnership with Boehringer Ingelheim (BI), leadXpro generated protein and provided the X-ray structure data to advance their drug discovery programs on CCR2, a key target in several autoimmune and neurological disorders.
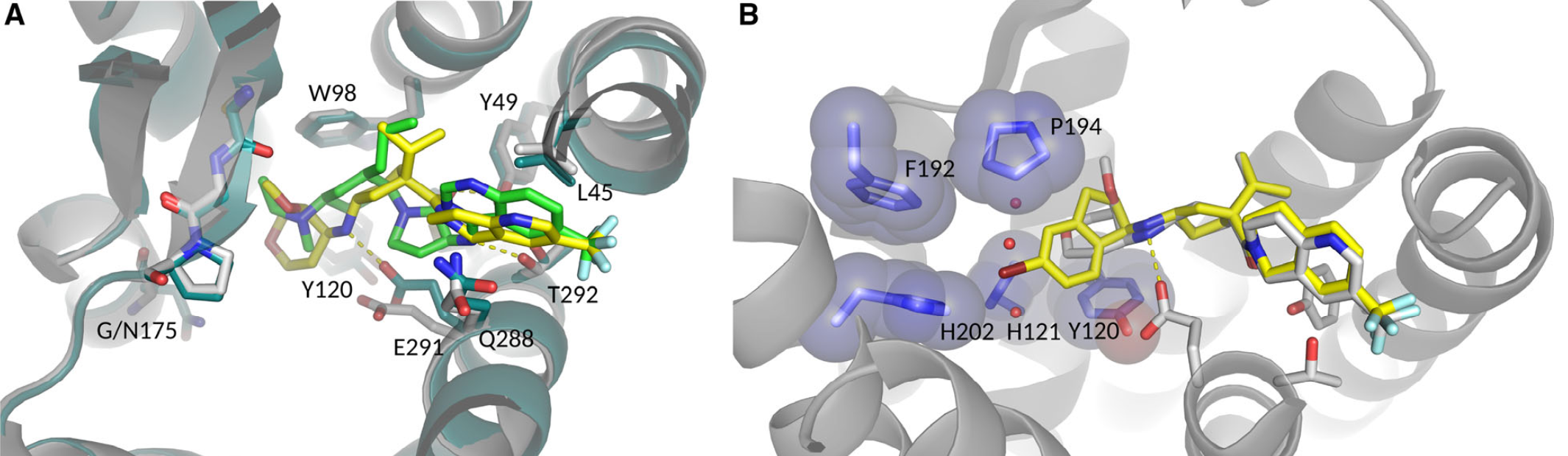
After extensive construct screening and optimization, the team at leadXpro were able to produce a 2.7 Å crystal structure of CCR2 with bound antagonist – this breakthrough came through the use of in meso in situ serial X-ray crystallography (IMISX), a method that is especially useful when working with fragile membrane protein crystals (1,3). The structural data aided Boehringer’s chemistry efforts to optimize drug residence time (2).
References
1 Apel, A.-K. et al. Crystal Structure of CC Chemokine Receptor 2A in Complex with an Orthosteric Antagonist Provides Insights for the Design of Selective Antagonists. Structure, doi:10.1016/j.str.2018.10.027 (2018).
2 Magarkar, A., Schnapp, G., Apel, A.-K., Seeliger, D. & Tautermann, C. S. Enhancing Drug Residence Time by Shielding of Intra-Protein Hydrogen Bonds: A Case Study on CCR2 Antagonists. ACS Medicinal Chemistry Letters 10, 324-328, doi:10.1021/acsmedchemlett.8b00590 (2019).
3 Cheng, R., Huang, C.-Y., Hennig, M., Nar, H. & Schnapp, G. In situ crystallography as an emerging method for structure solution of membrane proteins: the case of CCR2A. FEBS J. 287, 866-873, doi:10.1111/febs.15098 (2020).
Icon from (3)